News
Press Release – September 30, 2019
With recent EPA and DOE validation, Monarch Waste poised to disrupt multi-billion dollar US medical waste destruction market with PyroMed 550™ technology.
Monarch Waste Technologies, LLC (the “Company” or “Monarch”) with offices in Dallas, Texas and Albuquerque, New Mexico, was founded in 2015 by David Cardenas and Kevin Yearout. The Company recently announced that it has secured a Pyrolysis designation from the U.S. Environmental Protection Agency (“EPA”) and Department of Energy (“DOE”) that will enable the expansion of the company and its technology to develop and operate other locations across the United States.
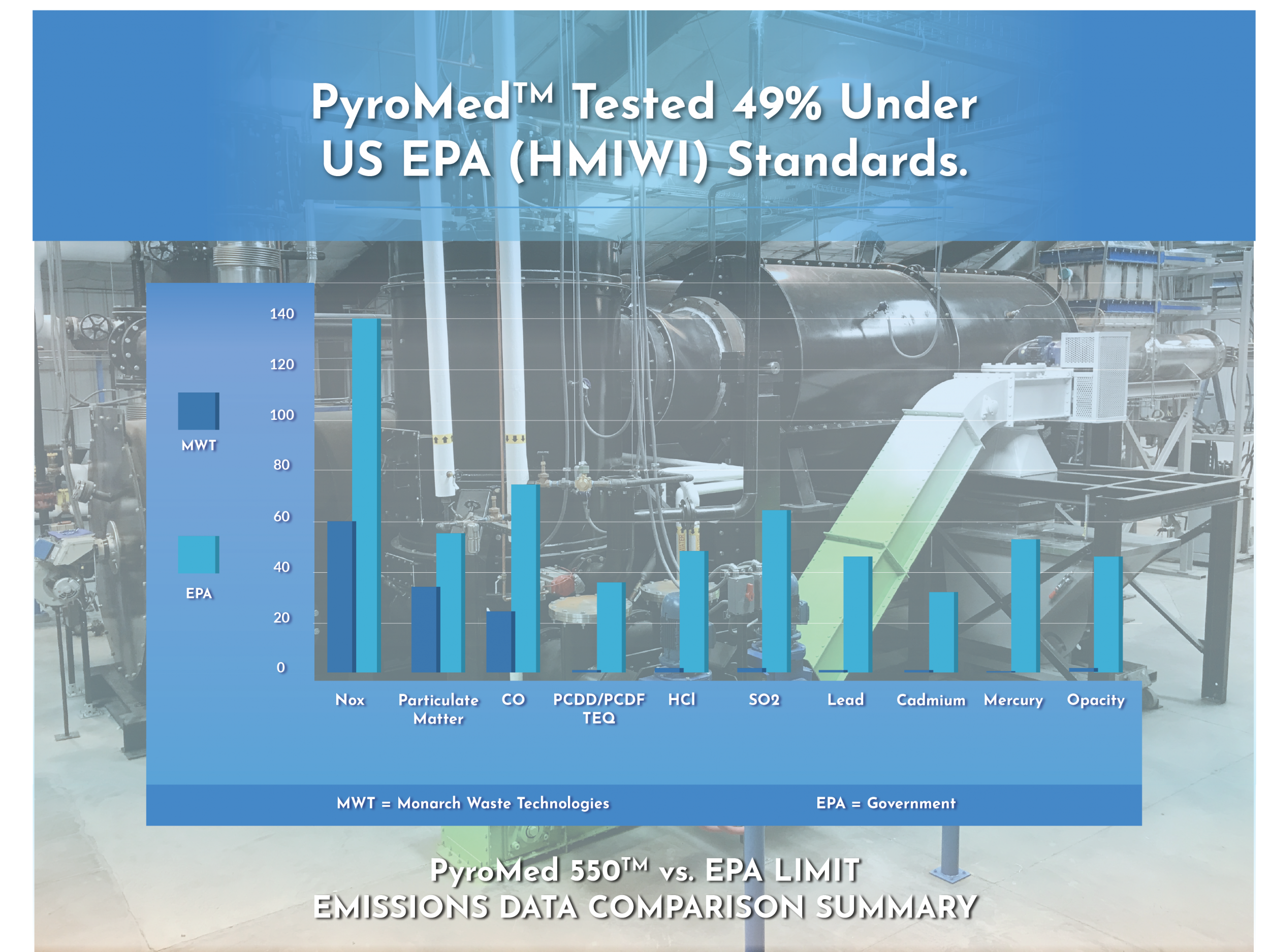
Monarch commissioned its first location in 2017 on tribal property outside of Santa Fe, New Mexico. After undergoing and successfully completing extensive federal permitting requirements, the technology (PyroMed 550TM) met EPAs most stringent test conditions and emissions limits (HMIWI Subpart Ec). In demonstrating the “worst-case scenario” emissions to the EPA, Monarch was required to use the most virulent waste mix possible in order to test the final emissions. The PyroMed 550TM achieved 49% cleaner emissions compared to EPA’s Ec emissions limits and a revealing 95% cleaner than EPA’s Subpart EEE limits allowed for hazardous waste combustors.
In 2016, prior to commissioning the unit, Monarch started an interactive process working closely with the EPA Region 6 and the administrators office in Washington to determine whether the PyroMed 550TM fit the definition of Pyrolysis (not Incineration). Extensive testing by Department of Energy (DOE) experts at Sandia National Labs in Albuquerque was performed to validate Pyrolysis.
The DOE issued a final report that validated the PyroMed 550TM was Pyrolysis and stated “…Monarch Waste system is consequently anticipated to significantly reduce emissions of harmful atmospheric pollutants compared with incineration…”
The EPA followed the DOE effort, and after multiple site visits to validate functional aspects of the unit, the EPA issued a final letter to Monarch on June 2019 that stated “…evidence is sufficient to state that the system [PyroMed 550TM] is a pyrolysis unit and produces primarily pyrolysis products, not products of combustion…”.This letter went on to determine that the unit would not be defined as a hospital, infectious, and medical waste incinerator (HIMWI) by EPA definition. This designation will allow the regulatory permitting process for other locations to be more efficient in time and cost. The federal EPA has elected to give a special exemption for pyrolysis as outlined within 40 CFR 60 and 62 rulings regarding incineration of hazardous/ infectious medical waste.
A Company official said “…Our first goal was to get the facility built and our system operational so we could actually show hospitals it works. Because up to that point, it was all pictures and concepts, nothing tangible to show. Our next goal was to secure governmental validation and testing to lay the path for quicker permitting which took more time than we anticipated but considering the impact of what we will accomplish from here, it was worth the effort. Now we are focused on speed to market and timing could not be better to expand the technology to major cities across the United States…”
Monarch’s approach to build, own, and operate small scale facilities closer to the initial location where the waste is generated has now developed from concept to reality. The company is in a position to revolutionize the process of how the healthcare sector destroys medical and hazardous waste. Their environmentally friendly PyroMed 550TM system is the future of safely dealing with medical waste and the conclusion from the DOE report gives evidence to say the PyroMed 550TM can solve this need.
The Company official further added “… we are in full development and permitting mode on several sites in strategic locations to serve one of our large customers who has operations across the entire United States. We are also finalizing negotiations with an existing customer to execute a long term contract to develop, own, and operate a facility for their large hospital system in the Midwest to replace their recently decommissioned incinerator with our PyroMed 550TM unit as well…”
The US Medical Waste Issue:
In 1995 the EPA inventory for Medical waste Incinerators listed over 2800 facilities in the US. Today there are only 8 remaining with no proven, validated or bankable options to consider as the remaining facilities continue their struggle to meet EPA standards. Medical waste levels are on the rise as healthcare expands with the rising population. Hospitals are under enormous strain to find permanent economical clean solutions to solve the issues of growing landfills and the effect that their waste is adding to them.
Definition of Pyrolysis
Pyrolysis is defined as the chemical decomposition induced in material using an endothermic process (external indirect heat) within an oxygen depleted environment. As waste continually enters the inside of the PyroMed proprietary sealed unit, the waste absorbs the heat, slowly “roasts / decomposes” but never ignites or combusts since it is impossible to do so in an oxygen depleted environment within the PyroMed closed loop process.
The PyroMed 550TM
Although the PyroMed 550TM is the first of its kind in the U.S. it is not a prototype or something new. The basic design has over 20 years of operating history on two other continents, which demonstrates the technology is both proven and bankable. Monarch owns the Intellectual Property (IP) with its partners in the UK. The prior design was improved with added IP to ensure compliance with EPA medical and hazardous waste standards. An added benefit of the process is generating “free” thermal heat. This heat is used to run the unit continually without the need of outside fuel sources. The system is self-sufficient once it is in operation, unlike most other systems which require natural gas or propane.
The excess heat can be used to create steam or can be diverted to a steam turbine to convert directly to electricity. The PyroMed 550TM is the only commercially proven continuous [not batch] medical waste process with this much operational history that is actually working in the United States today.
For more information on Monarch Waste Technologies, the PyroMed 550TM or investment opportunities contact either:
David Cardenas
davidc@monarchwastetechnologies.com
Kevin Yearout
keviny@monarchwastetechnologies.com
EPA Stack Tests February 2019 (49% under)
EPA Issued Determination Letter

DOE Determination / Report – November 12, 2018


